 
Its conductivity improves as it is heated. At room temperature, boron is a poor electrical conductor.Boron forms stable covalent bonds rather than ionic bonds. 
Boron can transmit part of infrared light.It can be used to produce green fire and is added as a common colorant in fireworks. Element atomic number 5 burns with a green flame.Because it is an excellent neutron absorber, it is used in nuclear reactor control rods. Boron is added to steel and other alloys. Boron is used in super hard materials, magnets, nuclear reactor shielding, semiconductors, to make borosilicate glassware, in ceramics, insecticides, disinfectants, cleaners, cosmetics, and many other products.It is a derivative of streptomycin and is called boromycin. The amorphous phase of boron is reactive, while crystalline boron is not reactive.Boron appears to have been formed by nuclear fusion from cosmic ray collisions, before the solar system was formed. While many elements are produced via nuclear fusion inside stars, boron is not among them.Crystalline boron is the second hardest element, following carbon.Boron has the highest melting point and boiling point of the metalloids.In 1909 Ezekiel Weintraub isolated nearly pure element number 5. In 1808, boron was partially purified by Sir Humphry Davy and also by Joseph L.Element atomic number 5 is rare in the solar system. Yet, pure boron is extremely difficult to produce and the element abundance is only 0.001% of the Earth's crust. Over a hundred minerals contain boron and it is found in several compounds, including boric acid, borax, borates, kernite, and ulexite.Element number 5 is present in the cell walls of all plants, so plants, as well as any animal that eats plants, contain boron.Boron exhibits properties of either metals or nonmetals, depending on the conditions.Eleven isotopes, with masses 7 to 17 are known. Natural boron consists of a mix of two stable isotopes: boron-10 and boron-11. A boron atom has 5 protons and 5 electrons.The word was used to describe borax, one of the boron compounds known to ancient man. The element name boron comes from the Arabic word buraq, which means white.Boron compounds form the basis for the classic slime recipe, which polymerizes the compound borax.The valence electrons are held closer towards the nucleus of the atom. This means that the nucleus attracts the electrons more strongly, pulling the atom's shell closer to the nucleus. The effect of increasing proton number is greater than that of the increasing electron number therefore, there is a greater nuclear attraction. However, at the same time, protons are being added to the nucleus, making it more positively charged. This is because, within a period or family of elements, all electrons are added to the same shell. 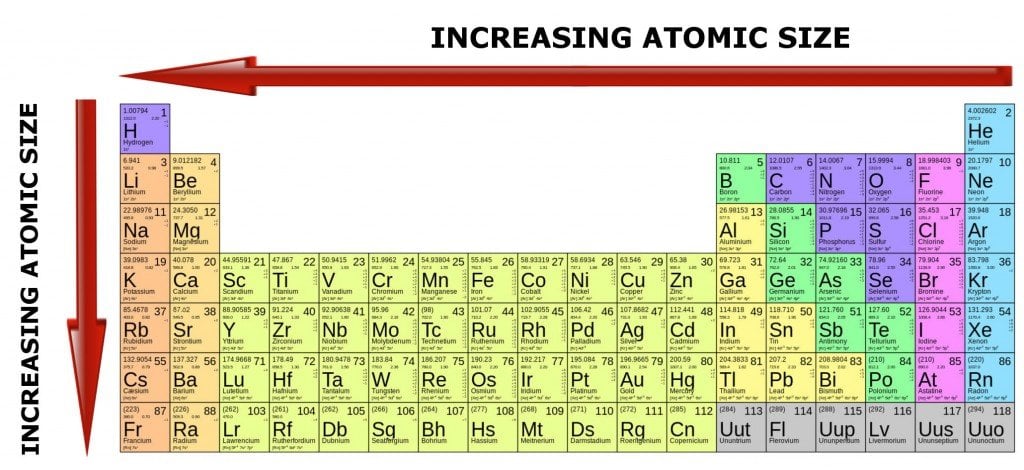
Atomic radius patterns are observed throughout the periodic table.Ītomic size gradually decreases from left to right across a period of elements. The covalent radii of these molecules are often referred to as atomic radii. Nevertheless, it is possible for a vast majority of elements to form covalent molecules in which two like atoms are held together by a single covalent bond. Some are bound by covalent bonds in molecules, some are attracted to each other in ionic crystals, and others are held in metallic crystals. However, this idea is complicated by the fact that not all atoms are normally bound together in the same way. The atomic radius is one-half the distance between the nuclei of two atoms (just like a radius is half the diameter of a circle). This is caused by the increase in atomic radius.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
